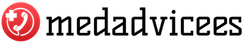 Fondaparinux sodium( trade name Arikstra) is a medical preparation in the form of a clear, colorless solution. It is an anticoagulant of direct action and a selective factor Xa inhibitor.
Fondaparinux sodium( trade name Arikstra) is a medical preparation in the form of a clear, colorless solution. It is an anticoagulant of direct action and a selective factor Xa inhibitor.
This medication is available as a solution for subcutaneous administration. It is released in syringes, each containing 5 mg of sodium prinoprino and additional components:
- sodium chloride;
- water for injection;
- hydrochloric acid;
- sodium hydroxide.
Contents
- Mechanism of action
- Mechanism of action
- Indications and contraindications for use
- Instruction for use
- Overdose and side effects
- Special instructions
- Interaction with other drugs
- Is the opinion. ..
- Purchase and storage of medication
Pharmacological profile
Aricstra is a pentasaccharide that is obtainedsynthetically. It is able to bind selectively to antithrombin III, thus catalyzing the inactivation of the factor Xa of the homeostasis system.
Due to the inactivation, the thrombus formation process breaks off at the thrombin generation stage. The use of the drug does not affect the fibrinolytic activity and the duration of hemorrhage.
Fondaparinux has no effect on platelets and does not cause various cross-reactions. The therapeutic dose of the drug does not change the results of such coagulation tests, such as APTT, ABC and MNO.In rare cases, when taking an excessive dose of the drug( more than 2.5 mg), the APTT is lengthened.
After subcutaneous inoculation, the drug is completely absorbed from the injection site. The maximum saturation of the drug in the blood plasma is observed two hours after its administration. 
The elimination half-life of the drug is twenty-five minutes after the inoculation. Most of the drug is localized in the blood and only a small amount is in the extravenous fluid.
The active ingredient of the drug binds to the AT III protein by 94%.With other plasma proteins, Fondaparinux binds in a small amount.
In vivo conditions, the metabolism of the drug has not been studied, since during normal operation of the kidneys, the medication is excreted in an unconverted form along with the urine.
In a healthy body, about 75% of the drug is excreted by the kidneys for 72 hours.
Mechanism of action
The antithrombotic activity of the drug is the consequence of selective inhibition of factor Xa. The mechanism of action of the drug consists in partial binding to the protein AT III, thereby increasing the ability to neutralize factor Xa.
As a result of neutralization, thrombus formation and thrombin are inhibited. The active ingredient does not inactivate thrombin and does not affect platelets.
Indications and contraindications for use
Arikstru is used in the following cases:
- for the prevention of thromboembolic complications in those patients who underwent severe orthopedic operations on the
 lower limbs;
lower limbs; - presence of severe coronary syndrome, which is manifested by periodic angina or myocardial infarction;
- pulmonary artery occlusion;
- acute deep vein thrombosis;
- vein occlusion therapy in non-surgical patients;
- acute pathognomic syndrome of superficial veins of lower extremities, with absence of blockage of deep veins;
- treatment of acute pulmonary embolism.
The drug is contraindicated in the following cases:
- active clinical hemorrhage;
- severe microbial endocarditis;
- high sensitivity to the constituent components of the drug;
- is a severe stage of kidney deficiency.
With special care, the drug is prescribed for the following diseases and conditions:
- high probability of hemorrhage;
- congenital or acquired destruction of the blood coagulation system( bleeding);
- pitting diseases of the stomach and duodenum in an acute degree;
- previously suffered from intracranial hemarthrosis;
- recently conducted ophthalmic surgery;
- patients who are more than 75 years old.
Instructions for Use
The use of the medication should only be carried out under the strict supervision of a physician. The drug is used by subcutaneous or intravenous inoculation. Places for subcutaneous administration are the left and right anterolateral surface of the facial abdominal wall.
 In case of intravenous inoculation, the drug essence is dialed into a catheter or into a container with sodium chloride( 0.9%), in which it is necessary to first dilute the drug.
In case of intravenous inoculation, the drug essence is dialed into a catheter or into a container with sodium chloride( 0.9%), in which it is necessary to first dilute the drug.
After the injection, the catheter is washed with a large amount of saline.
In case of using mini-containers, the infusion is carried out for 1-2 minutes.
The dosages of the drug depend on the therapeutic purposes:
- Venous obstructive complications .The recommended dose of the drug is 2.5 mg with a multiplicity of inoculation once a day. The course of treatment is carried out throughout the phase of high risk of venous thromboembolism( at least six to nine days).For patients who underwent surgical interventions after fractures of the bones, the course of treatment with the drug 10-15 days.
- PE and deep vein thrombosis .The dose of Arikstra for patients with a weight of 50-100 kg is 7.5 mg. Patients weighing more than 100 kg are injected subcutaneously with 10 mg of the drug solution. Therapy is carried out for six to seven days and stops when the patient is transferred to oral anticoagulants.
- Periodic angina and myocardial infarction without enlargement of the ST segment .The prescribed dose of the drug is inoculated subcutaneously, in an amount of 2.5 mg once daily. Treatment is carried out for eight days.
How to get a shot in the stomach:
Overdose and side effects
An overdose of a medicine can cause bleeding. In this case, it is necessary to cancel the administration of the drug. To stop bleeding surgical hemostasis, plasmapheresis and transfusion of fresh blood plasma are used.
Possible side effect:
- hematopoietic system : hemorrhage( including intracerebral and intracranial), thalassemia, thrombocytopenia, blood clotting disorder, platelet anomaly;
- metabolism : hypokalemia;
- digestive system : vomiting, nausea, gastritis, indigestion;
- respiratory system : shortness of breath, cough( in rare cases).
Special instructions
During the use of the drug in order to prevent complications after surgery, it is necessary to adhere exactly to the time during which the inoculation of the initial dose of the drug occurs( after six hours after the operation)  .
.
In case of impaired renal function, medication is prescribed with caution, since there is a possibility of hemorrhage development. The same applies to the use of the drug in severe destruction of liver function.
With regard to the use of the drug in childhood, the administration of 0.1 mg of the drug per kg of body weight is based on the same exposure as in adults with the treatment of deep vein thrombosis.
Arikstra is not prescribed during childbearing and during lactation.
Interaction with other drugs
Clinical studies have shown that the combined use of fondaparinux sodium with antiplatelet agents, oral anticoagulants and cardiac glycosides has no effect on its pharmacodynamics and pharmacokinetics.
Arikstru is not recommended to be mixed with other medications, as there is no data on their compatibility.
There is an opinion. ..
Reviews of practicing doctors, as well as their patients who took the drug Arikstra.
Very effective drug, but with improper application it can cause bleeding. I appoint it only after a careful examination of patients and strictly follow the process of injection.
Antonina Mikhailovna
Arikstra really has a positive effect, but the only drawback is that it has side effects. I do not recommend using it myself without prescribing and monitoring the doctor.
Evgeniy Anatolievich
I was pricked with this drug, because I was diagnosed with acute thrombosis. There were no side effects, but the state of health was not very good. I was administered 2.5 mg of the drug once a day.
Elena
My husband was stabbed with this medicine four times( he got into an accident and did several operations to him).But half a year has already passed and he is constantly headache and there are dizziness.
I consulted with a neurologist, I thought it might be such a long-term side effect, but he said that this can not be. Therefore, we do not even know what it could be.
Angelina
It can be seen from the experience of using the drug that in most cases it has the necessary effect, but many side effects may appear.
Purchase and storage of medication
Arikstra is produced in the form of a solution and dispensed in syringes, the average price of the medication is 5000-6000 rubles. 
The drug should be stored at a temperature of no more than 25 degrees in a place inaccessible to children.
Freezing solution is not recommended. The drug is dispensed only by prescription.
Analogues of Arikstra medication:
- Pentosan polysulphate ( injection solution in 1-ml ampoules);
- and direct - Fondaparinux sodium .