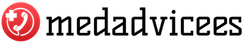- General description
- Where is
- Useful properties
- Drugs based on it
- Contraindications
- Harm
General characteristic
Sodium benzoate E211( the Latin name sodium benzoate) is a salt of benzoic acid and has the properties of a preservative. The chemical formula is C6H5COOH.The substance is a white powder, odorless and tasteless.
It was discovered in the XIX century by a scientist named Hugo Fleck. For many years he tried to find a substitute for hard-to-reach salicylic acid, and in 1875 he did it. However, it was only in 1908 that the additive was used for its intended purpose, that is, for preserving products.
Where
is contained In nature, the preservative E211 is contained in:
- apples;
- prune;
- ;
- cranberry;
- mustard;
- cinnamon;
- carnations and other products.
But basically, a food supplement is obtained in the laboratory.
Very often sodium benzoate is added to:
- confectionery;
- sauces, ketchups;
- margarine, mayonnaise;
- caviar and canned food;
- carbonated sweet drinks;
- non-alcoholic beer and alcohol;
- packaged juices and nectars;
- jujube, jam, jams.
Useful properties
In Russia and CIS countries the substance is actively used in various fields of production. In the food industry, sodium benzoate is used for canning, since it slows the action of enzymes. It also gives the products a sweetish flavor and enhances color.
Sodium benzoate is used in the manufacture of cosmetics, pyrotechnics and in medicine. Its solution acts as an expectorant.
Preparations based on it
Thanks to its properties, the preservative is a part of very many medicines. The substance is contained in hydrogen peroxide.
Very popular now Caffeine-sodium benzoate, it is sold in tablets or ampoules and is dispensed without a doctor's prescription. It perfectly helps when burning fat in bodybuilding, so it is actively used for weight loss.

It is important to strictly dose the drug according to the instructions, otherwise the risk of side effects increases.
to contents ^Contraindications
Harm
- Under the influence of sodium benzoate, redox reactions are much worse, which can cause allergies, and in the worst case - cirrhosis, kidney problems, Parkinson's disease or even cancer.
- Interaction with vitamin C turns the formulation into benzene, which is a dangerous carcinogen.
- Intelligence, nervous system and behavior of adults and children will have negative consequences in the form of a decrease in the intelligence level, hyperactivity, development or strengthening of attention deficit disorder when eating foods that combine sodium benzoate and any of the following dyes( E102, E104, E110, Е122, Е124, Е129).
- The substance is fatal for all kinds of felines.
Scientists do not recommend eating foods containing preservative E211, so as not to cause irreparable harm to health.